
Oxygen segregation to a grain boundary in molybdenum
A. R. Waugh and M. J. Southon
An imaging atom-probe field-ion microscope together with a time-of-flight spectrometer has been used here to show the spatial distribution of oxygen atoms at a grain boundary in molybdenum.
This picture shows field ion images of molybdenum and oxygen ions from a grain boundary in molybdenum, where oxygen has segregated. Most of the oxygen is located within a band 1 nm width at the grain boundary. The concentration of oxygen atoms at the boundary is about 1018 atoms per square metre. The segregation of oxgyen makes the boundaries in molybdenum brittle, leading to intergranular failure. For more details, see Waugh, A. R., and M. J. Southon. "Surface studies with an imaging atom-probe." Surface Science 68 (1977): 79-85.
Photographs courtesy of Dr Sally Waugh.
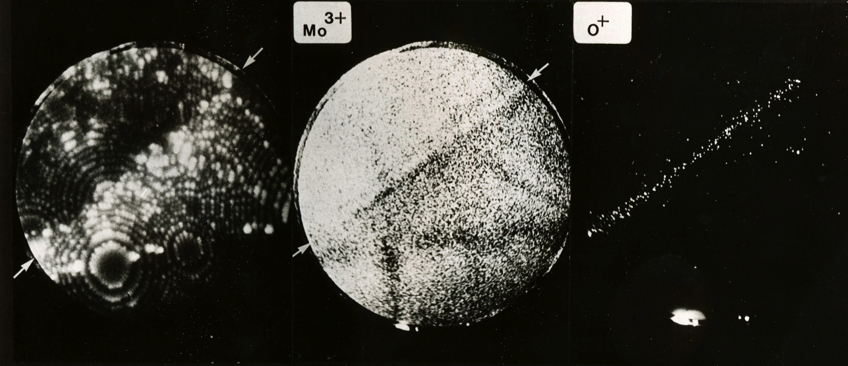
Segregation of oxygen atoms to molybdenum grain boundary. Each dot represents an atom. Note that the grain boundaries are visible in the Mo3+ image because of an associated local change in the curvature of the specimen surface that leads to a corresponding local change in magnification.
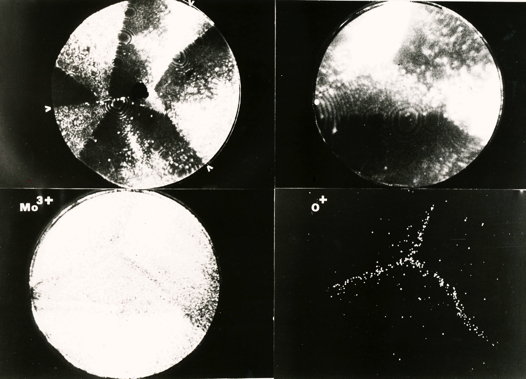
Segregation of oxygen atoms to molybdenum grain boundary triple point. Each dot represents an atom.
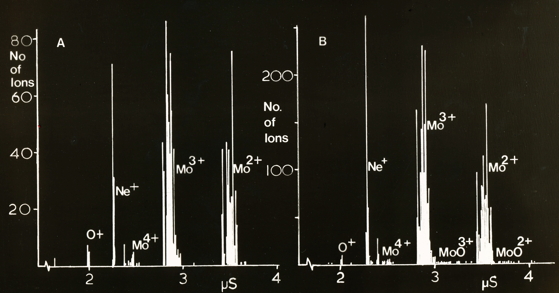
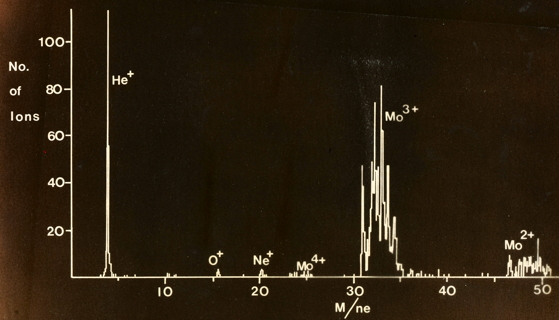
Ion spectra recorded on the time-of-flight spectrometer
|
|
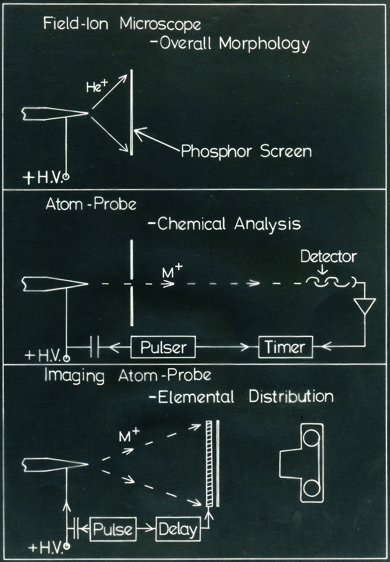
Schematic illustration of the field ion microscope, atom probe and imaging atom probe. "HV" stands for high voltage.

Part of the actual instrument built by Bob Waugh and Mike Southon, at the University of Cambridge, Department of Metallurgy.

Part of the actual instrument built by Bob Waugh and Mike Southon, at the University of Cambridge, Department of Metallurgy.
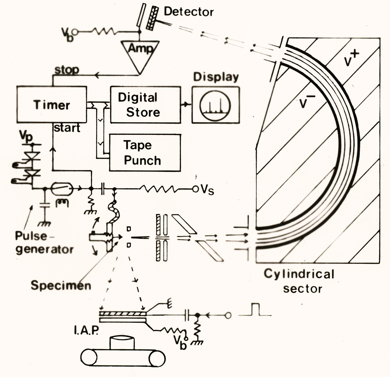
Schematic illustration of the field ion microscope, atom probe and time-of-flight mass spectrometer. "I.A.P." stands for imaging atom probe, Note the paper-tape punch used to record data.

The cylindrical sector illustrated schematically above, as it actually appears. This is a part of the time-of-flight mass spectrometer set.
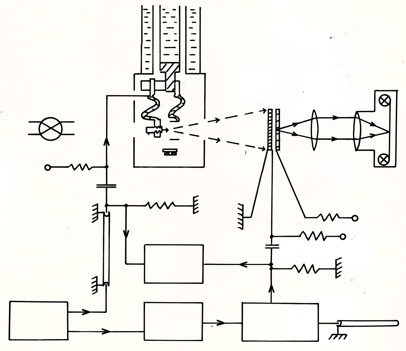
Circuit diagram. The sample is kept at liquid helium temperature.
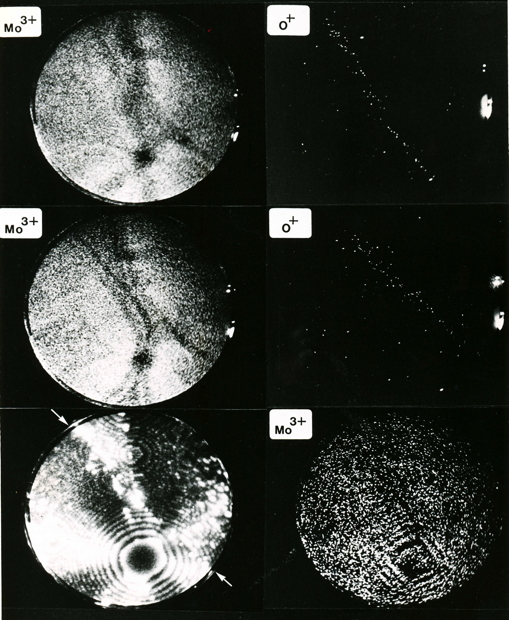
Segregation of oxygen atoms to a grain boundary in molybdenum.














