Commentary: lengthening kinetics of Widmanstätten ferrite
This is a useful and in some respects, inspiring publication: Arya, Nabeel, Zurob, Phillion. Materials Today Communications 50 (2026) 114488.
Measurements are reported of the lengthening rate of Widmanstätten ferrite plates, as revealed by the transformation-induced invariant-plane strain displacements during confocal laser scanning microscopy (resolution 1.3 μm). This is for a steel of chemical composition Fe-0.12C-0.018Si-2.07Mn wt%, austenitised at 1200°C, then transformed during continuous cooling at 3 K s-1. The steel has a calculated Ae3=796°C.
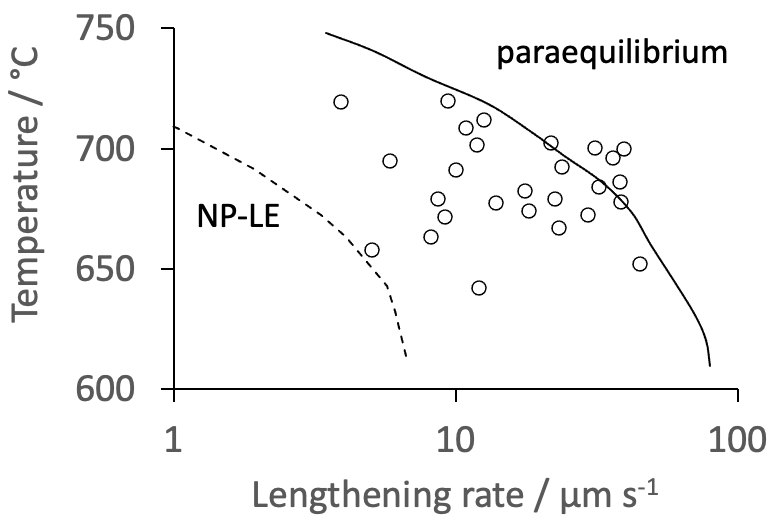
Figure 1 shows the results. I have chosen to exclude all measurements other than for plates originating from the austenite grain surfaces and secondary plates from allotriomorphic ferrite. This is because in my judgement, intragranular nucleation cannot be stereologically established from two-dimensional observations unless the austenite grain size is massive. Nucleation from "existing plates" is excluded given the limited resolution of the method, making it difficult to detect contact between parallel plates. These exclusions do not affect any discussion because the data fall in the same range illustrated, as can be seen in the original publication by Arya et al. The scatter in the data is most likely due to the difficulty of ensuring observations of plates growing parallel to the observation surface, as explained by Arya et al. It is normal in these circumstances to assume that the largest plates observed are representative of the true growth rate.
Note that the measured data are not isothermal but are plotted against temperature presumably using average values over the temperature ranges involved.
To analyse the growth rates, the authors use an empirical representation of Trivedi's theory [1], fitted by Hillert [2]: \begin{equation}\frac{v_\text{max} r_\text{c}}{\overline D_\text{C}^\gamma} =\frac{1}{4} \frac{\Omega_\circ}{1-\Omega_\circ} \times 10^{-2.5}(1-\Omega_\circ) \end{equation} where \(\Omega_\circ\) is a supersaturation, \(r_\text{c}\) the critical plate tip radius at which growth cannot occur, and there is a weighted average carbon diffusion coefficient \(\overline D_\text{C}^\gamma\) according to Trivedi. It is assumed that the interfacial energy per unit area is 0.2 J m-2 and that the plate adopts a tip-radius that leads to a maximum in the growth rate.
The growth original study compared the data against paraequilibrium calculations, and assuming a negligible-partitioning local-equilibrium (NP-LE) condition due to Coates [3], where even Mn maintains equilibrium at the interface and hence has to diffuse, inconsistent with the observed displacive transformation mechanism - the confocal microscopy relies on those displacements.
The width of the concentration spike of manganese from the NP-LE estimates was not included in the paper. This is important, because a gradient energy term in fact makes such transformation impossible [4]. There is no experimental evidence ever produced to show the concentration spike. It is likely that the NP-LE concept is best abandoned if the widths of the substitutional solute spikes are not physically meaningful [5].
The data in Figure 1 are well explained by the paraequilibrium growth model, whereby Widmanstätten ferrite is a carbon diffusion-controlled displacive transformation. To quote Christian [6], "Considering an alloy in which the two components occupy different sets of sites, as for example in an interstitial solid solution, it may happen that, at the transformation temperature, one component is sufficiently mobile for long range diffusion to take place, while the other is immobile. The whole transformation may then consist in the diffusion of the mobile component .... combined with a rearrangement of the atomic configuration of the other component by a martensitic type transformation."
The agreement shown in Figure 1 with paraequilibrium estimates, was demonstrated in 1985 [7]. There is, therefore, no need to invoke a mythical barrier [6] to interfacial motion, and I am pleased that the idea has been rejected implicitly by the authors. Interfaces associated with displacive transformations are glissile (french glisser to slip, slide + english ile).
Further thoughts
Continuous cooling versus isothermal growth
Simulating a growing parabolic cylinder (plate) under non-isothermal conditions adds complexity because the growth-solution assumes a steady state that is constantly being disrupted by the shifting boundary conditions. When the temperature \(T\) drops, the local-equilibrium solute concentrations (determined by the phase diagram) shift, changing the supersaturation and the concentration gradient driving the growth. The solute profile in the matrix may not keep up with the shifting interface concentration.
A steady-state approximation where the concentration at the interface is changed stepwise in a numerical scheme [8] will not be representative. The profile of concentration around the plate at \(T_1>T_2\) is not, in that scenario, inherited in successive steps such as at \(T_2\). That original profile will reduce concentration gradients in the next step, so anisothermal growth will in general be slower than an isothermal growth experiment conducted at the same temperature.
The critical plate tip radius \(r_\text{c}\) will not be identical at all temperatures because it is driving force dependent.
The blue curve in the video below shows the slower anisothermal growth, whereas the red curve indicates what would happen during isothermal growth. These calculations are simply intended to be illustrative.
NP-LE
The application of the hypothetical negligible-partitioning local-equilibrium model of transformation to plate growth, oddly allows for capillarity (Gibbs-Thompson) correction for carbon, but not for manganese, which also diffuses. And since the fluxes of the two solute concentrations have to be linked to maintain local-equilibrium, this should also influence the carbon concentration at the plate tip. I don't know whether this will be a significant effect, but this needs to be established in the context of the application of NP-LE to curved interfaces.
Diffusional-displacive
There is an unfortunate use of the term diffusional-displacive in the context of Widmanstätten ferrite.
Plates can be precipitated with the same shape deformation as martensite, yet inherit a different substitutional solute content relative to the parent phase. The precipitates have a coherent or semi-coherent interface that permits a lattice correspondence to be maintained between the parent and product phases. The diffusion that facilitates the composition change requires atoms to move readily over the sites predicted by the lattice correspondence without creating additional sites or destroying existing sites by vacancy migration [9,5]. Any other diffusion would locally modify the shape change.
There is no suggestion anywhere that Widmanstätten ferrite has a substitutional-atom to iron-atom ratio that is different from the average in the steel. Its association with this term is not justified. The type of reaction where diffusional-displacive is descriptive, is, for example, when silver and aluminium in solid solution combine to precipitate Ag2Al, i.e., a gross change in chemical composition.
References
- Trivedi, Metallurgical Transactions 1 (1970) 921-927.
- Hillert, Metallurgical Transactions A 6(1975) 5-19.
- Coates, Metall Trans 4 (1973) 395-396.
- Bhadeshia, Current Opinion in Solid State and Materials Science 20 (2016) 396-400.
- Bhadeshia, Metallurgical and Materials Transactions A 57 (2026) 1-20.
- Christian, Physical Props. of Martensite & Bainite, Iron & Steel Institute, London, 1965, 1-19 ISI special report 56.
- Bhadeshia, Materials Science and Technology 1 (1985) 497-504.
- Bhadeshia, Svensson, Gretoft, Acta Metallurgica 33 (1985) 1271-1283.
- Christian, Progress in Materials Science 42 (1997) 101-108.
