Coupled Diffusional/Displacive Transformations
Proceedings of an International Conference: Phase Transformations '87,
Institute of Metals, London,
Edited by G. W. Lorimer, 1988, pp. 322-325, by G.B. Olson, H.K.D.H. Bhadeshia and M. Cohen
This scientific paper investigates the kinetics of phase transformations in steels,
specifically focusing on the transition from austenite (γ)
to ferrite (α). The authors propose a mathematical model
that integrates diffusional and displacive mechanisms to calculate how quickly an
interface moves during the growth of plates.
By analysing how free energy is dissipated through both solute diffusion and
interface motion, the study establishes a method for determining transformation
velocities across various temperatures. The research highlights that while
carbon partitioning occurs during certain transformations, others proceed
without composition changes, particularly below the martensite start
temperature (Ms).
Ultimately, this framework provides a unified theoretical basis for understanding
the formation of martensite, bainite, and Widmanstätten ferrite.
Download publication
Audio podcast
Slides
Study Guide: Coupled Diffusional and Displacive Transformations
This study guide examines the theoretical and experimental analysis of phase transformations in crystals involving both substitutional and interstitial elements, specifically focusing on the γ → α transformation in iron-carbon alloys. The document synthesises the relationship between diffusion-controlled and interface-controlled growth mechanisms.
Part I: Short-Answer Quiz
Instructions: Answer the following ten questions in 2–3 sentences based on the provided source context.
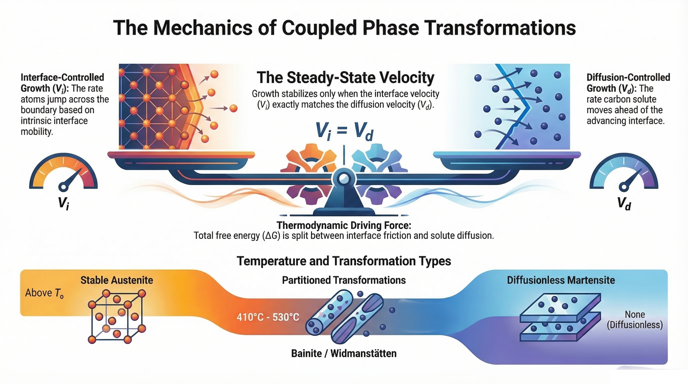
1. What are the two primary processes that dissipate the net free energy (ΔG) available for interface motion?
2. Define the condition required to find the actual growth velocity (V) of a moving interface.
3. What does the term Gsurf represent in the context of phase transformations?
4. According to the Ivantsov equation, what factors determine the term Vd (diffusion-controlled velocity)?
5. How is the motion of the α/γ interface physically modelled in this study?
6. What is the calculated martensite start temperature (Ms) for an Fe-0.4C wt.% alloy according to the source?
7. Describe the trend of the growth rate (Vmax) as the transformation temperature decreases from 530°C to 410°C.
8. How do bainite and Widmanstätten ferrite nucleation differ from martensite nucleation regarding solute partitioning?
9. What is the significance of the Péclét number (p) in the calculations?
10. Explain why the nucleation velocity passes through a maximum (forming a C-curve) while growth velocity does not.
Part II: Answer Key
1. What are the two primary processes that dissipate the net free energy (ΔG) available for interface motion?
The net free energy is dissipated by the interface process (Gi), which relates to the intrinsic mobility of the interface, and the diffusion process (Gd), which involves the redistribution of alloying elements partitioned ahead of the moving interface.
2. Define the condition required to find the actual growth velocity (V) of a moving interface.
The actual velocity is determined by the intersection of two functions: Vi = ξ{Gi} and Vd = ψ{Gd}. The steady-state velocity occurs when the velocity calculated from interface mobility exactly equals the velocity computed from the diffusion of solute ahead of the interface.
3. What does the term Gsurf represent in the context of phase transformations?
Gsurf is the energy stored during the creation of a new interface area and the energy required for shape deformation during the phase change. It is not considered part of the energy dissipated by the motion itself but is a prerequisite energy cost that reduces the free energy available for the transformation.
4. According to the Ivantsov equation, what factors determine the term Vd (diffusion-controlled velocity)?
Vd is determined by the carbon concentration in the austenite ahead of the interface (x̄ - xγ) relative to the concentration difference across the interface (xγ - xα), as well as the weighted average diffusivity of carbon (D̄) and the effective tip radius of the transforming plate (ρ).
5. How is the motion of the α/γ interface physically modelled in this study?
The interface motion is modelled using the theory for thermally activated dislocation motion adapted for interfacial structures. This involves calculating the Gibbs free energy of activation (G*) and considering the maximum glide resistance (Ḡid) presented by obstacles to the interface.
6. What is the calculated martensite start temperature (Ms) for an Fe-0.4C wt.% alloy according to the source?
The Ms temperature for this specific alloy is calculated to be 397°C. This temperature marks the point below which diffusionless martensitic transformations occur.
7. Describe the trend of the growth rate (Vmax) as the transformation temperature decreases from 530°C to 410°C.
The growth rate (Vmax) increases monotonically as the temperature decreases within this range. While the diffusivity of carbon in austenite decreases with lower temperatures, the increase in driving force at higher undercooling compensates for it, resulting in a higher overall growth velocity.
8. How do bainite and Widmanstätten ferrite nucleation differ from martensite nucleation regarding solute partitioning?
Bainite and Widmanstätten ferrite nucleate at temperatures above Ms, where the driving force is insufficient for diffusionless transformation; therefore, they involve solute partitioning where the carbon concentration in the embryo (xα) is less than the bulk concentration (x̄). Martensite nucleation, occurring below Ms, involves no such partitioning.
9. What is the significance of the Péclét number (p) in the calculations?
The Péclét number is a dimensionless velocity given by p = Vρ/2D̄. It is a critical parameter in the Ivantsov equation used to relate the interface velocity and the diffusion of solute.
10. Explain why the nucleation velocity passes through a maximum (forming a C-curve) while growth velocity does not.
Nucleation involves a higher Gsurf (stored energy) term compared to growth, which makes the interface velocity more sensitive to the decreasing diffusivity of carbon at lower temperatures. This creates a competition between increasing driving force and decreasing mobility, resulting in a maximum velocity at an intermediate temperature.
Part III: Essay Questions
- The Partitioning of Free Energy: Discuss how the available free energy (ΔG) is distributed between the interface process and the diffusion process. Explain how the "maximum velocity criterion" helps determine the level of carbon supersaturation in the growing ferrite.
- Kinetics of Displacive Transformations: Compare the temperature dependence of nucleation versus growth in Fe-0.4C alloys. Why does nucleation exhibit C-curve kinetics while growth shows a monotonic increase in velocity as temperature drops toward the Ms?
- The Role of Solute Diffusion: Analyse the importance of the weighted average diffusivity (D̄) of carbon in austenite. How does the carbon concentration gradient at the α/γ interface influence the steady-state velocity of the transformation?
- Modelling Interface Mobility: Describe the application of thermally activated dislocation motion theory to the study of interfacial movement. What are the roles of the Gibbs free energy of activation (G*) and the activation volume (V*) in this model?
- Classification of Transformations: Based on the source context, explain the theoretical transition between martensite, bainite, and Widmanstätten ferrite. Focus on how the degree of carbon supersaturation (xα/x̄) changes with transformation temperature and driving force.
Part IV: Glossary of Key Terms
| Term |
Definition |
| Bainite |
A transformation product occurring at temperatures above Ms that involves displacive nucleation with solute partitioning. |
| D̄ (Weighted Average Diffusivity) |
The average diffusion coefficient of carbon in austenite, calculated by integrating the concentration-dependent diffusivity D{x, T} over the concentration range ahead of the interface. |
| Displacive Transformation |
A phase change involving the coordinated movement of atoms, often modelled by the motion of an interfacial dislocation structure. |
| Gd |
The free energy dissipated during the diffusion process of solute ahead of the moving interface. |
| Gi |
The free energy dissipated by the interface process, relating to the resistance of the interface to motion. |
| Gsurf |
The stored energy required for the creation of new interface area and shape deformation during a phase transformation. |
| Ivantsov Equation |
A mathematical relation used to calculate the diffusion-controlled velocity of a curved, needle-like or disc-like particle. |
| Ms Temperature |
The martensite start temperature; the temperature below which the driving force is sufficient for completely diffusionless transformation. |
| Péclét Number (p) |
A dimensionless constant representing the ratio of the product of velocity and tip radius to the diffusivity (Vρ/2D̄). |
| Solute Partitioning |
The redistribution of alloying elements (like carbon) between the parent phase and the new phase during transformation. |
| Widmanstätten Ferrite |
A transformation product similar to bainite that forms via displacive nucleation at relatively high temperatures with low supersaturation. |
| xα |
The mole fraction of carbon in the newly formed α phase (ferrite/martensite). |
| xγ |
The mole fraction of carbon in the γ phase (austenite) at the α/γ interface. |

Published 2021















