Chromium carbide (Cr23C6) and M23C6 in steels
H. K. D. H. Bhadeshia
Thermodynamic and crystallographic analysis of chromium carbides,
specifically the M23C6 phase, and their critical
role in enhancing creep-resistant steels used in power plants. The research explores how the
addition of boron and iron influences the stability and coarsening of these carbides, which is
essential for maintaining structural integrity under high-temperature stress.
By utilising first-principles calculations and the FLAPW method, the
authors estimate formation enthalpies and examine how boron substitution can decrease the energy
of the system to stabilise the carbide structure.
Included are detailed crystal structure models, X-ray diffraction data, and experimental
evidence of boron's distribution within the steel's matrix. Ultimately, this work aims to bridge
the gap between macroscopic measurements and atomistic theory to develop more durable alloys
for energy infrastructure.
Audio podcast
The crystallographic data on which the information below is based are from A. L. Bowman, G. P. Arnold, E. K. Storms and N. G. Nereson,
Acta Crystallographica B28 (1972) 3102-3103.
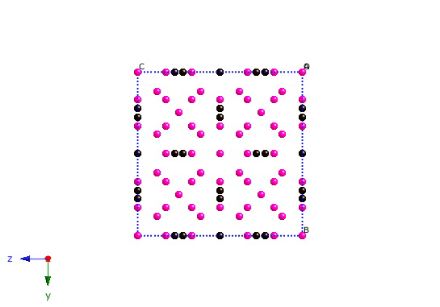
Cell projected along [100]. Carbon coloured black.
|
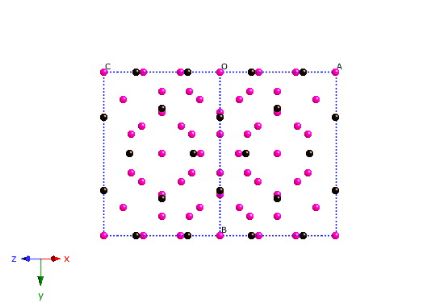
Cell projected along [110].
|
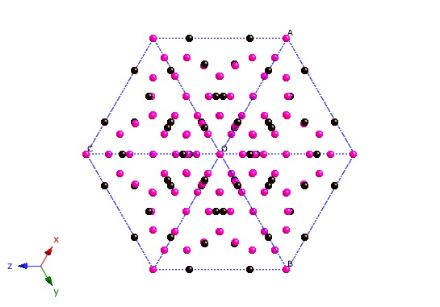
Cell projected along [111].
|
|
Zone Axis Projections

[001] zone axis |
 |

[110] zone axis |
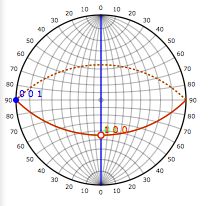 |

[111] zone axis |
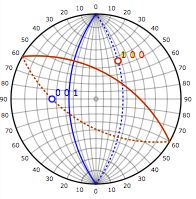 |

[112] zone axis |
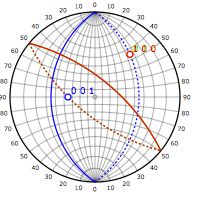 |
X-ray Diffraction
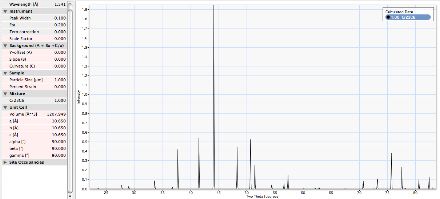
Unlabelled X-ray diffraction pattern |
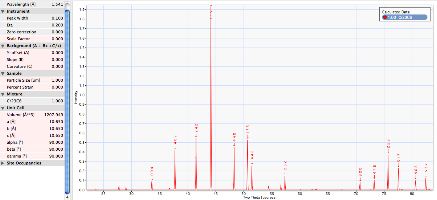
Annotated X-ray diffraction pattern |
Study guide
This guide is an overview of the structural, thermodynamic, and mechanical properties of chromium carbide.
and the effects of boron and iron substitution within creep-resistant steels. This research may be useful for the development of alloys used
in high-temperature power plant applications.
Part I & II: Quiz and Answer Key
1. What is the crystal structure and space group of ?
possesses a Cubic-F crystal structure belonging to the
space group.
The unit cell contains 92 chromium atoms distributed across four symmetry sites (4a, 8c, 32f, 48h) and 24 carbon atoms located at the 24e site.
2. Define the three stages of the creep deformation process.
Creep consists of primary (transient) creep, where the creep rate decreases due to strain hardening;
secondary (steady-state) creep, where the rate remains constant due to a balance between hardening and recovery;
and tertiary creep, where the rate increases until failure or rupture occurs.
3. Why is the steady-state (secondary) creep rate a critical engineering design parameter for power plants?
Power plant components are designed for long service lives, often around 30 years, where failure is dangerous and difficult to control.
A decreased steady-state creep rate slows the overall deformation of the material, thereby increasing the total time to rupture.
4. How does the addition of boron influence the creep behaviour of 9–12 wt% chromium steels?
Small additions of boron remarkably increase creep rupture strength and decrease the creep rate by stabilising
carbides
and retarding their coarsening rate. Boron dissolves evenly within the
phase.
5. What is the observed effect of iron (Fe) substitution on the lattice parameter of ?
Substituting chromium with iron generally decreases the lattice parameter and the unit cell volume of the carbide.
For instance, calculations show the unit cell volume decreases from
in
to
in
.
6. What is the role of boron substitution in thermodynamic stability?
Boron substitution into carbon sites decreases the formation enthalpy of the carbide, making the structure more thermodynamically stable.
The formation enthalpy drops from
for
to
for
.
Part III: Essay Format Questions
- Crystallographic Orientation Relationships: Analyse the "cube-cube" relationship between
and austenite ().
- Boron as a Stabilising Agent: Evaluate the mechanism by which boron improves creep resistance.

Published 2021


























