
A discussion of diffusion and its effects can be found in a set of lectures available online.
Steel is incredibly versatile because it can be heat-treated in order to produce a vast range of microstructures and associated mechanical properties. The heat-treatment usually involves the steel being heated into a temperature in the austenite phase field. This temperature is quite high, in the range 800-1200 oC, depending on the details of the chemical composition.
Commercial heat treatments are generally carried out in electrical (resistance) furnaces or natural gas fired furnaces. The size of the furnace may range from a moderately sized building to handle many thousands of tonnes of steel, to a furnace the size of a microwave oven.
It is inevitable that the furnace atmosphere contains oxygen. More accurately, the chemical potential of carbon in the atmosphere may be lower than that in the steel being heat treated. Carbon will therefore be removed from the steel by the process commonly known as "Decarburization".
The chemical composition of steel M0 is approximately Fe-0.8C wt%. It has been heated in an electric furnace, without any particular protection, at 1200 oC for 2 hours and then cooled slowly to ambient temperature.
The sample of M0 was then ground flat on one surface using SiC grinding paper lubricated with water, followed by polishing with fine diamond paste. Once an acceptable polish was obtained, the sample was etched in 2% nital (a mixture of nitric acid and methanol) for 20 seconds before washing with methanol and drying using warm air. The resulting cross-section reveals the extent of decarburization at the surface, with remarkable changes in microstructure with distance away from the surface.

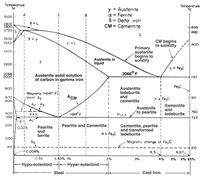
| Iron-carbon phase diagram, reproduced with the permission of Jud Ready of the Georgia Tech. Joint Student Chapter of ASM/TMS. |
You will find below a series of micrographs taken as a function of position away from the surface, and at a variety of magnifications. You can click on each image to see an expanded version. Additional information on decarburization and its commercial importance can be found in the links at the bottom of this page.
Decarburised surface (low magnification) .

| The microstructure away from the surface of the steel. There is a much smaller quantity of ferrite. |
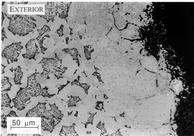
| Higher magnification image. The microstructure at the free surface of the steel. At the higher magnification it becomes clear that the top surface is almost completely denuded of carbon. The oxygen has therefore started to oxidise the iron. The oxidation is penetrating the prior austenite grain boundaries because these are high-energy sites. A much higher magnification image is also available to illustrate this phenomenon. |

| Higher magnification image. The microstructure away from the surface of the steel. The Widmanstatten ferrite is more apparent at this higher magnification. Notice that the steel cannot be of exactly the eutectoid composition since there is some ferrite even in the regions which are not decarburized. It is better described as hypoeutectoid. A much higher magnification image is also available to illustrate this phenomenon. |
Explain how decarburisation causes a variation in the microstructure in a slowly cooled eutectoid steel as a function of the distance from the exposed surface.
A Fe-0.7C wt.% steel is decarburised at 1200 K such
that a constant carbon concentration of 0.1 wt.% is maintained at the
exposed surface. If the diffusion coefficient for carbon in
austenite is
![]() , how long will it
take for the depth at which the concentration is 0.4 wt.% to become
2.5 mm?
, how long will it
take for the depth at which the concentration is 0.4 wt.% to become
2.5 mm?
How does your calculated time compare with an estimate made assuming that
the diffusion distance is ![]() ? Comment on why the two results
are different.
? Comment on why the two results
are different.
How can decarburisation be prevented in practice?
By referring to the phase diagram, it can be deduced that the surface will be rich in ferrite, the unaffected regions away from the surface will be fully pearlitic. The intermediate regions will have a mixture of ferrite and pearlite according to the lever rule applied at the eutectoid temperature.
Using the error function solution,
On the other hand, with
The difference arises because this estimate assumes random walk, whereas diffusion in a concentration gradient is driven along a particular direction.
Decarburisation can be prevented by heat treatment in an intert atmosphere, by wrapping the component in stainless steel foil, or by painting with an isolating paint.
| Superalloys | Titanium | Bainite | Martensite | Widmanstätten ferrite |
| Cast iron | Welding | Allotriomorphic ferrite | Movies | Slides |
| Neural Networks | Creep | Mechanicallly Alloyed | Theses | Retained Austenite |
| PT Group Home | Materials Algorithms |

|

|