Introduction
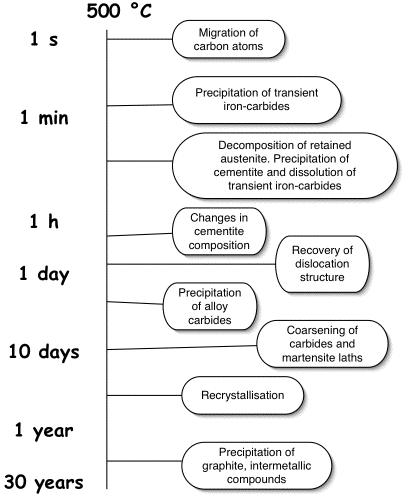
Tempering is a term historically associated with the heat treatment of martensite in steels. It describes how the microstructure and mechanical properties change as the metastable sample is held isothermally at a temperature where austenite cannot form. The changes during the tempering of martensite can be categorised into stages.
During the first stage, excess carbon in solid solution segregates to defects or forms clusters within the solid solution. It then precipitates, either as cementite in low-carbon steels, or as transition iron-carbides in high-carbon alloys. The carbon concentration that remains in solid solution may be quite large if the precipitate is a transition carbide. Further annealing leads to stage 2, in which almost all of the excess carbon is precipitated, and the carbides all convert into more stable cementite. Any retained austenite may decompose during this stage. Continued tempering then leads to the coarsening of carbides, extensive recovery of the dislocation structure, and finally to the recrystallisation of the ferrite plates into equiaxed grains.
This is a useful description but it is revealing to consider first, the factors responsible for driving the process in the first place.
Deviation from Equilibrium
Tempering is a process in which the microstructure approaches equilibrium under the influence of thermal activation. It follows that the tendency to temper depends on how far the starting microstructure deviates from equilibrium. It is interesting therefore to consider how metastable a material can be, before dealing specifically with martensite. Turnbull characterised metastability in terms of the unit RTm where R is the universal gas constant and Tm is the absolute melting temperature. This coarse unit is a measure of the thermal energy in the system at the melting temperature; it represents a large amount of energy, typically in excess of 20,000 J mol−1.
| Metastable structure | RTm |
|---|---|
| Supersaturated solutions | 1 |
| Amorphous metal | 0.5 |
| Modulated films, nanostructures | 0.1 |
Supersaturated solutions are prominent in this list and the extent of metastability depends both on the excess concentration and on the equilibrium solubility. It can be demonstrated that excess carbon which is forced into solution in martensite is the major contributor to the stored energy of martensite.
The calculations presented in Table 2 show the components of the stored energy of martensite in a typical low-alloy martensitic steel Fe-0.2C-1.5Mn wt%. It is necessary to define a reference state, which is here taken to be an equilibrium mixture of ferrite, graphite and cementite, with a zero stored energy. Graphite does not in fact form because it is too slow to precipitate; the effect of replacing the graphite with cementite is to increase the stored energy by some 70 J mol−1.
When transformations occur at low temperatures, it is often the case that substitutional elements like manganese and iron cannot diffuse during the time scale of the experiment, whereas carbon is still mobile. The transformation then happens in such a way that the Fe/Mn ratio is maintained constant whilst the carbon redistributes subject to this constraint, until its chemical potential becomes uniform. This is known as paraequilibrium. Unlike the equilibrium state, because the iron and manganese atoms are trapped during transformation, their chemical potentials are no longer uniform. This adds a further 315 J mol−1 to the stored energy.
When bainite forms, the transformation mechanism is displacive, there is a shape deformation, which leads to an additional 400 J mol−1 of stored energy. Since there is no diffusion during transformations, but the carbon partitions following growth, the total stored energy is that for the paraequilibrium state added to the strain energy term, giving a net value of 785 J mol−1.
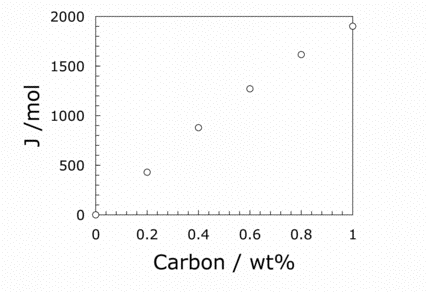
Martensite is not only a diffusionless transformation, but it frequently occurs at low temperatures where its virgin microstructure is preserved. Even the carbon remains trapped in the product crystal. Furthermore, the strain energy term associated with martensite is greater at about 600 J mol−1 because the plates tend to have a larger aspect ratio (thickness/length). There may also be twin interfaces within the martensite plates, which cost about 100 J mol−1. The trapping of carbon inside the martensite adds a further 629 J mol−1, which makes the total stored energy in excess of 1700 J mol−1!
| Phase Mixture in Fe-0.2C-1.5Mn wt% at 300 K | Stored Energy / J mol−1 |
|---|---|
| 1. Ferrite, graphite and cementite | 0 |
| 2. Ferrite and cementite | 70 |
| 3. Paraequilibrium ferrite and paraequilibrium cementite | 385 |
| 4. Bainite and paraequilibrium cementite | 385 + 400 = 785 |
| 5. Martensite | 385 + 600 + 100 + 629 = 1714 |
| 6. Mechanically alloyed ODS metal | 55 |
Figure 1: The free energy due to the trapping of carbon in martensite, as a function of its carbon concentration. The results are for a temperature of 473 K.
Virgin Microstructure
The virgin microstructure obtained immediately after quenching from austenite consists of plates or laths of martensite which is supersaturated with carbon. In the vast majority of steels, the martensite contains a substantial density of dislocations which are generated during the imperfect accommodation of the shape change accompanying the transformation. The plates may be separated by thin films of retained austenite, the amount of untransformed austenite becoming larger as the martensite-start temperature MS is reduced.
Carbon Atoms
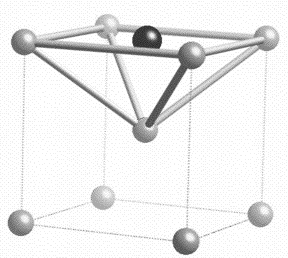
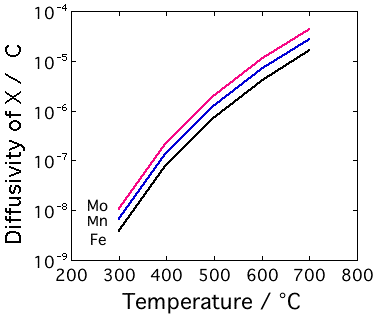
Carbon is an interstitial atom in ferritic iron, primarily occupying the octahedral interstices. There are three such interstices per iron atom. At a typical concentration of 0.4 wt% or about 2 at%, less than 1% of these interstices are occupied by carbon. Furthermore, there is a strong repulsion between carbon atoms in nearest neighbour sites. This means that carbon atoms almost always have an adjacent interstitial site vacant, leading to a very high diffusion coefficient when compared with the diffusion of substitutional solutes. In the latter case, the substitutional vacancy concentration is only 10−6 at temperatures close to melting, and many orders of magnitude less at the sort of temperatures where martensite is tempered. It follows that carbon diffuses much faster than substitutional atoms (including iron), as illustrated below.
Precipitation of Iron Carbides
In high-carbon steels, the precipitation of excess carbon begins with the formation of a transition carbide, such as ε (Fe2.4C). ε-carbide can grow at temperatures as low as 50 °C. Indeed, most of the iron carbides can precipitate at low temperatures, well below those associated with the motion of substitutional solutes. This is because they grow by a displacive mechanism which does not require the redistribution of substitutional atoms (including iron); carbon naturally has to partition. This corresponds to a process known as paraequilibrium transformation in which the iron to substitutional solute ratio is maintained constant but subject to that constraint, the carbon achieves a uniform chemical potential.
Martensite is said to be supersaturated with carbon when the concentration exceeds its equilibrium solubility with respect to another phase. However, the equilibrium solubility depends on the phase. The solubility will be larger when the martensite is in equilibrium with a metastable phase such as ε carbide. Some 0.25 wt% of carbon is said to remain in solution after the precipitation of ε-carbide is completed.
Although most textbooks will begin a discussion of tempering with this first stage of tempering, involving the redistribution of carbon and precipitation of transition carbides, cementite can precipitate directly. This is particularly the case when the defect density is large. Trapped carbon atoms will not precipitate as transition carbides but cementite is more stable than trapped carbon.

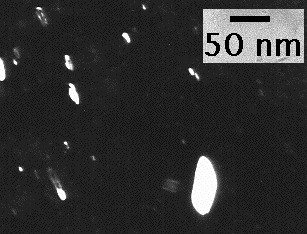
(a) Transmission electron micrograph of martensite in a Fe-4Mo-0.2C wt% steel after tempering at 190 °C for 1 hour. The carbon has in this case precipitated as fine particles of cementite. (b) Corresponding dark-field image showing the distribution of retained austenite, which has not been affected by the tempering.
Decomposition of Retained Austenite

Tempering at higher temperatures, in the range 200–300 °C for 1 h induces the retained austenite to decompose into a mixture of cementite and ferrite. When the austenite is present as a film, the cementite also precipitates as a continuous array of particles which have the appearance of a film.
Dark field transmission electron micrograph of martensite in a Fe-4Mo-0.2C wt% steel after tempering at 295 °C for 1 hour. Only the cementite is illuminated. The film of cementite at the martensite plate boundaries is due to the decomposition of retained austenite.
Further Tempering
Tempering at even higher temperatures leads to a coarsening of the cementite particles, with those located at the plate boundaries growing at the expense of the intra-plate particles. The dislocation structure tends to recover, the extent depending on the chemical composition. The recovery is less marked in steels containing alloying elements such as molybdenum and chromium.
Bright field transmission electron micrograph of martensite in a Fe-4Mo-0.2C wt% steel after tempering at 420 °C for 1 hour.

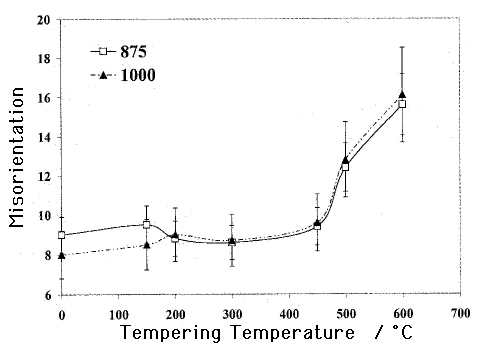
The recovery of the dislocation structure and the migration of dislocation-cell and martensite boundaries leads not only to a coarsening of the plates, but also an increase in the crystallographic misorientation between adjacent plates, as illustrated in the adjacent figure. The data are from Suresh et al., Ironmaking and Steelmaking 30 (2003) 379–384.
Precipitation of Alloy Carbides
Alloy carbides include M2C (Mo-rich), M7C3, M6C, M23C6 (Cr-rich), V4C3, TiC etc., where the 'M' refers to a combination of metal atoms.
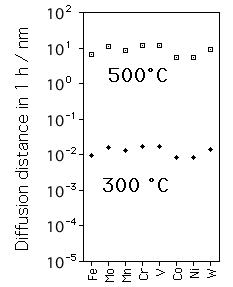
However, all of these carbides require the long-range diffusion of substitutional atoms. They can only precipitate when the combination of time and temperature is sufficient to allow this diffusion. The figure on the left shows the calculated diffusion distance in ferrite for a tempering time of 1 h. It is evident that the precipitation of alloy carbides is impossible below about 500 °C for a typical tempering time of 1 h; the diffusion distance is then just perceptible at about 10 nm.
The alloy carbides grow at the expense of the less stable cementite. If the concentration of strong carbide forming elements such as Mo, Cr, Ti, V, Nb is large then all of the carbon can be accommodated in the alloy carbide, thereby completely eliminating the cementite.


Fe-0.1C-1.99Mn-1.6Mo wt% quenched to martensite and then tempered at 600 °C. (Photograph courtesy of Shingo Yamasaki). The precipitates are needles of Mo2C particles. The needles precipitate with their long directions along ⟨100⟩α.
Severe Tempering

Fe-0.98C-1.46Si-1.89Mn-0.26Mo-1.26Cr-0.09V wt% tempered at 730 °C for 7 days. This transmission electron micrograph shows large cementite particles and a recovered dislocation substructure. The plate microstructure is coarsened but nevertheless retained because the carbides are located at plate boundaries.
Hardness
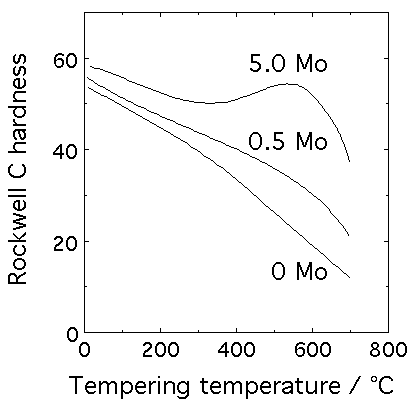
Fe-0.35C-Mo alloy quenched to martensite and then tempered at the temperature indicated for one hour (data from Bain's Alloying Elements in Steels). Whereas the plain carbon steel shows a monotonic decrease in hardness as a function of tempering temperature, molybdenum in this case leads to an increase in hardness once there is sufficient atomic mobility to precipitate Mo2C.
Secondary hardening is usually identified with the tempering of martensite in steels containing strong carbide forming elements like Cr, V, Mo and Nb. The formation of these alloy carbides necessitates the long-range diffusion of substitutional atoms and their precipitation is consequently sluggish. Carbides like cementite therefore have a kinetic advantage even though they may be metastable. Tempering at first causes a decrease in hardness as cementite precipitates at the expense of carbon in solid solution, but the hardness begins to increase again as the alloy carbides form. Hence the term secondary hardening.
Kinetics
Case Studies: AerMet 100
AerMet 100 is a martensitic steel which is used in the secondary-hardened condition; its typical chemical composition is as follows:
| C | Co | Ni | Cr | Mo | Mn | Si | Al | Ti | S | P |
|---|---|---|---|---|---|---|---|---|---|---|
| 0.23 | 13.4 | 11.1 | 3.0 | 1.2 | 0.03 | 0.03 | 0.004 | 0.013 | 0.001 | 0.003 |
The cobalt plays a key role in retarding the recovery of martensite during tempering, thereby retaining the defect structure on which M2C needles can precipitate as a fine dispersion. By increasing the stability of body-centred cubic iron, it also reduces the tendency of martensite to revert to austenite during tempering. The carbon concentration is balanced such that all the cementite is replaced by the much finer alloy carbides during secondary hardening.
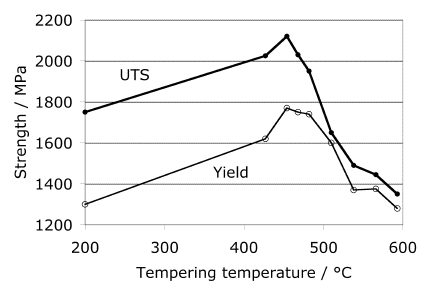
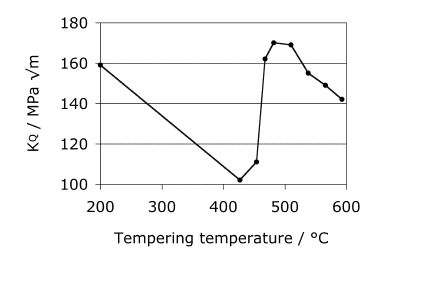
The optimum combination of strength and toughness is obtained by tempering at 470 °C. Tempering at 430 °C, 5 h is associated with a minimum in toughness because of the precipitation of relatively coarse cementite platelets in a Widmanstätten array.
Landing gear critical components are made from tempered martensite.




Temper Embrittlement
Tempering can lead to embrittlement when the steel contains impurities such as phosphorus, antimony, tin and sulphur. These impurities tend to segregate to the prior austenite grain boundaries and reduce cohesion across the boundary plane, resulting in intergranular failure.
Role of Prior Austenite Grain Boundaries
Ordinary steels are ferritic or pearlitic; both of these phases can grow by reconstructive transformation across austenite grain boundaries. By contrast, the coordinated motion of atoms accompanying displacive transformations cannot be sustained across austenite grain boundaries. Therefore, a vestige of the austenite grain boundary (prior austenite grain boundary) remains in the microstructure.
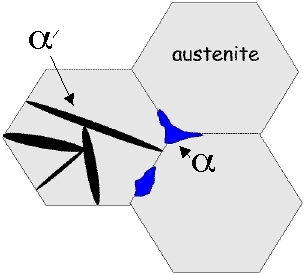

Figure: (a) Martensite (α') is hindered by austenite grain boundaries whereas allotriomorphic ferrite (α) is not. (b) Failure at the prior austenite grain boundary.
650 °C Reversible Temper Embrittlement
Tempering at temperatures around 650 °C promotes the segregation of impurity elements such as phosphorus to the prior austenite grain boundaries. The tendency for embrittlement correlates strongly with an empirical J factor:
J = Mn + Si + 104 (P + Sn)
where the concentrations of elements are in weight percent.
To summarise, the impurity-controlled temper embrittlement occurs in bainite as it does in martensite; after all, neither of these transformation products cross austenite grain surfaces and hence leave them open for impurity segregation. By comparison, reconstructive transformation products such as allotriomorphic ferrite, can grow across and consume the austenite grain surfaces, thereby removing them entirely from the final microstructure.
Finally, it is worth noting that although the science of the embrittlement is well understood, for reasons of cost, commercial steels always contain more impurities than is desirable. Steps must therefore be taken to mitigate the impurity effects, for example by alloying with molybdenum to pin down the phosphorus and prevent it from segregating.
300–350 °C Temper Embrittlement
Fracture is again intergranular with respect to the prior austenite grain boundaries which become decorated with coarse cementite particles during tempering. At the same time, the grain boundaries are weakened by impurity segregation. The cementite particles crack under the influence of an applied stress and in this process concentrate stress at the weakened boundaries. These factors combine to cause embrittlement.
300–350 °C Tempered-Martensite Embrittlement
This effect is common in clean steels, with fracture occurring transgranularly relative to the prior austenite grain boundaries. It is attributed to the formation of cementite particles at the martensite lath boundaries and within the laths. During tempering, the particles coarsen and become large enough to crack, thus providing crack nuclei which may then propagate into the matrix. As a consequence, untempered low-carbon martensitic steels sometimes have a better toughness than when they are tempered, even though the untempered steel is stronger. The cementite behaves like a brittle inclusion.
Both of the impurity-controlled embrittlement phenomena can be minimised by adding about 0.5 wt% molybdenum to the steel. The Mo associates with phosphorus atoms in the lattice thereby reducing mobility and hence the extent to which they segregate to boundaries. Larger concentrations of molybdenum are not useful because precipitation occurs.
In many bainitic microstructures, tempering even at temperatures as high as 550 °C has only a small effect on cementite size and morphology. Consequently, the low-temperature embrittlement phenomena are not found in conventional bainitic microstructures.
Related Topics
- Tempered martensite in Fe-Mo-C
- Tempered martensite in Fe-V-C
- Tempered martensite in Fe-V-Mo-C
- Tempered martensite in Fe-9Cr-1Mo
- More about martensite.
Martensitic transformations
- Short review of martensite crystallography and nucleation
- Comprehensive book on martensite crystallography
- Elementary undergraduate lecture on martensite
- Slightly more advanced undergraduate lecture on martensite
- Tempered martensite
- Research publications on martensite
- Crystallography of austenite, ferrite and interstices
- Bain correspondence and Bain strain
- Deformation due to martensitic transformation
- Deformation due to martensitic transformation: interference microscopy
- Quench distortion
- Morphology and Crystallography.
Transformation hardening
Movies
- Shape memory effect.
- Magnetic shape memory effect.
- Model of displacive transformation.
- Growth of bainite.
- Videos of lectures on martensite

Tempered martensitic steel band holding log in position.

Tempered martensitic steel band holding log in position.

Lucerne, Switzerland

Lucerne, Switzerland